Page 49 - Abbott
P. 49
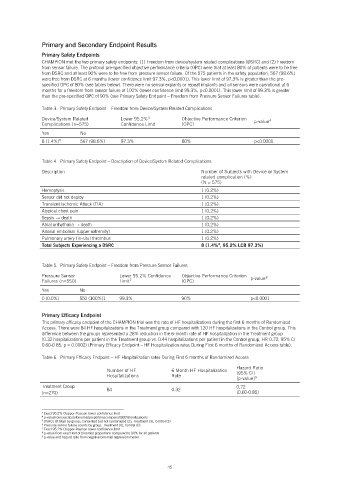
Primary and Secondary Endpoint Results
Primary Safety Endpoints
CHAMPION met the two primary safety endpoints: (1) Freedom from device/system related complications (DSRC) and (2) Freedom
from sensor failure. The protocol pre-specified objective performance criteria (OPC) were that at least 80% of patients were to be free
from DSRC and at least 90% were to be free from pressure sensor failure. Of the 575 patients in the safety population, 567 (98.6%)
were free from DSRC at 6 months (lower confidence limit 97.3%, p<0.0001). This lower limit of 97.3% is greater than the pre-
specified OPC of 80% (see tables below). There were no sensor explants or repeat implants and all sensors were operational at 6
months for a freedom from sensor failure of 100% (lower confidence limit 99.3%, p<0.0001). This lower limit of 99.3% is greater
than the pre-specified OPC of 90% (see Primary Safety Endpoint – Freedom from Pressure Sensor Failures table).
Table 3. Primary Safety Endpoint – Freedom from Device/System Related Complications
3
Device/System Related Lower 95.2% Objective Performance Criterion p-value
4
Complications (n=575) Confidence Limit (OPC)
Yes No
5
8 (1.4%) 567 (98.6%) 97.3% 80% p<0.0001
Table 4. Primary Safety Endpoint – Description of Device/System Related Complications
Description Number of Subjects with Device or System
related complication (%)
(N = 575)
Hemoptysis 1 (0.2%)
Sensor did not deploy 1 (0.2%)
Transient Ischemic Attack (TIA) 1 (0.2%)
Atypical chest pain 1 (0.2%)
Sepsis → death 1 (0.2%)
Atrial arrhythmia → death 1 (0.2%)
Arterial embolism (upper extremity) 1 (0.2%)
Pulmonary artery (in-situ) thrombus 1 (0.2%)
6
Total Subjects Experiencing a DSRC 8 (1.4% , 95.2% LCB 97.3%)
Table 5. Primary Safety Endpoint – Freedom from Pressure Sensor Failures
Pressure Sensor Lower 95.2% Confidence Objective Performance Criterion p-value
8
7
Failures (n=550) Limit (OPC)
Yes No
0 (0.0%) 550 (100%)1 99.3% 90% p<0.0001
Primary Efficacy Endpoint
The primary efficacy endpoint of the CHAMPION trial was the rate of HF hospitalizations during the first 6 months of Randomized
Access. There were 84 HF hospitalizations in the Treatment group compared with 120 HF hospitalizations in the Control group. This
difference between the groups represented a 28% reduction in the 6-month rate of HF hospitalization in the Treatment group
(0.32 hospitalizations per patient in the Treatment group vs. 0.44 hospitalizations per patient in the Control group, HR 0.72, 95% CI
0.60-0.85, p = 0.0002) (Primary Efficacy Endpoint – HF Hospitalization rates During First 6 months of Randomized Access table).
Table 6. Primary Efficacy Endpoint – HF Hospitalization rates During First 6 months of Randomized Access
Hazard Ratio
Number of HF 6 Month HF Hospitalization
Hospitalizations Rate (95% CI)
9
[p-value]
Treatment Group 84 0.32 0.72
(n=270) (0.60-0.85)
3 Exact 95.2% Clopper-Pearson lower confidence limit
4 p-valuefromexacttestofbinomialproportionscomparedto80%forallpatients
5 DSRCs (8 total) by group: Consented but not randomized (2), Treatment (3), Control (3)
6 Pressure sensor failure counts by group: Treatment (0), Control (0)
7 Exact 95.2% Clopper-Pearson lower confidence limit
8 p-value from exact test of binomial proportions compared to 90% for all patients
9 p-value and hazard ratio from negative binomial regression model
45