Page 51 - Abbott
P. 51
Freedom from Death or First HF Hospitalization
The proportion of patients who died or had at least one HF hospitalization in the Treatment group (44.8%) was smaller than in the
Control Group (51.8%) with a relative risk reduction of 23% (HR 0.77, 95% CI 0.60 – 0.98).
All Cause Hospitalizations
All cause hospitalizations were reduced in the Treatment group (554 in the Treatment group vs. 672 in the Control group, HR 0.84,
95% CI 0.75 – 0.95). The NNT per year to prevent one all cause hospitalization was 4. For every 100 patients treated, 26 all cause
hospitalizations would be prevented per year.
Death or All Cause Hospitalizations
Death or all cause hospitalizations were reduced in the Treatment group (604 in the Treatment group vs. 736 in the Control group,
HR 0.84, 95% CI 0.76 – 0.94). The NNT per year to prevent one death or all cause hospitalization was 4. For every 100 patients
treated, 29 deaths or all cause hospitalizations would be prevented per year.
Results from the Open Access Period (Longitudinal Analysis)
In the Open Access period, physicians were given access to PA pressures in the Control group for the first time while access to PA
pressures for the Treatment group continued. In the Control group, new physician access to PA pressures resulted in a 48%
reduction in HF hospitalization rates (0.36 vs. 0.68, HR 0.52, 95% CI 0.40-0.69, p<0.0001*). In the Treatment group, continued
physician access to PA pressures resulted in the maintenance of low HF hospitalization rates (0.45 vs. 0.48, HR 0.93, 95% CI 0.70-
1.22, p=0.5838*).
To account for potential longitudinal confounders, the change in HF hospitalization rates in the Control group as result of new access
to PA pressures was compared to the change in HF hospitalization rates in the Treatment group. The change in HF hospitalization
rates in the Control group was significantly greater than in the Treatment group (HR 0.56, 95% CI 0.38-0.83, p=0.0040*), indicating
that the significant 48% reduction in HF hospitalization rates observed in the Control group was the result of physician access to PA
pressure and not longitudinal effects.
(*P-values should be interpreted with caution because the analyses including Part 2 data were not specified before the onset of the
study and there are various sources of confounding effects which cannot be separated from the treatment effect.)
Treatment Effects in Women
The CHAMPION study was not powered to show statistical significance for gender, thus a complete determination of the effect of the
device in women cannot be made. At the request of FDA, a post-hoc gender analysis was conducted for the CHAMPION study, and
the initial finding of a treatment-by-gender interaction for the effect of the CardioMEMS device on the HF hospitalization rate was
related to (1) fewer women being in the study and the short duration of follow-up leading to a small number of events in women; and
(2) the low HF hospitalization rate in Control women due to an early excess of deaths in women in the Control group, which acted as
a competing risk to censor the occurrence of hospitalizations for heart failure.
A further analysis of the treatment-by- gender interaction was performed over the full period of Randomized Access and by
incorporating death in the Cox Proportional Hazards. When these limitations and confounding issues were evaluated over the full
period, there was neither a qualitative nor quantitative treatment-by-gender interaction and the treatment effect remained positive,
independent of gender. However, the analyses conducted do not alleviate the possibility of a diminished treatment effect in women.
Given the small number of women enrolled and small number of events observed in women, the true treatment effect in this group
remains uncertain. In order to further complement and evaluate the results obtained during the CHAMPION study, the effect of the
device in women is being studied as part of the post approval study.
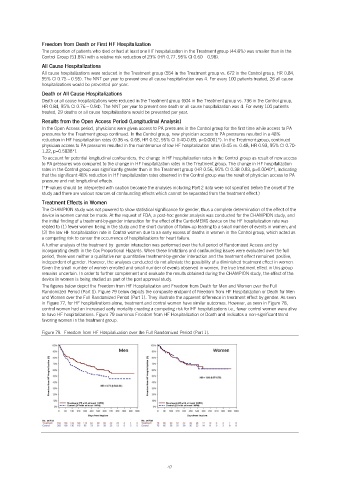
The figures below depict the Freedom from HF Hospitalization and Freedom from Death for Men and Women over the Full
Randomized Period (Part 1). Figure 79 below depicts the composite endpoint of Freedom from HF Hospitalization or Death for Men
and Women over the Full Randomized Period (Part 1). They illustrate the apparent difference in treatment effect by gender. As seen
in Figure 77, for HF hospitalizations alone, treatment and control women have similar outcomes. However, as seen in Figure 78,
control women had an increased early mortality creating a competing risk for HF hospitalizations i.e., fewer control women were alive
to have HF hospitalizations. Figure 79 examines Freedom from HF Hospitalization or Death and indicates a non-significant trend
favoring women in the treatment group.
Figure 79. Freedom from HF Hospitalization over the Full Randomized Period (Part 1).
47