Page 53 - Abbott
P. 53
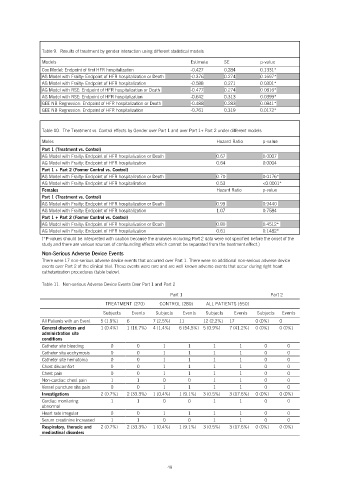
Table 9. Results of treatment by gender interaction using different statistical models
Models Estimate SE p-value
Cox Model: Endpoint of first HFR hospitalization -0.427 0.284 0.1331*
AG Model with Frailty: Endpoint of HFR hospitalization or Death -0.376 0.274 0.1697*
AG Model with Frailty: Endpoint of HFR hospitalization -0.588 0.271 0.0301*
AG Model with RSE: Endpoint of HFR hospitalization or Death -0.477 0.274 0.0816*
AG Model with RSE: Endpoint of HFR hospitalization -0.642 0.313 0.0399*
GEE NB Regression: Endpoint of HFR hospitalization or Death -0.488 0.283 0.0841*
GEE NB Regression: Endpoint of HFR hospitalization -0.761 0.319 0.0172*
Table 10. The Treatment vs. Control effects by Gender over Part 1 and over Part 1+ Part 2 under different models
Males Hazard Ratio p-value
Part 1 (Treatment vs. Control)
AG Model with Frailty: Endpoint of HFR hospitalization or Death 0.67 0.0007
AG Model with Frailty: Endpoint of HFR hospitalization 0.64 0.0004
Part 1 + Part 2 (Former Control vs. Control)
AG Model with Frailty: Endpoint of HFR hospitalization or Death 0.70 0.0176*
AG Model with Frailty: Endpoint of HFR hospitalization 0.53 <0.0001*
Females Hazard Ratio p-value
Part 1 (Treatment vs. Control)
AG Model with Frailty: Endpoint of HFR hospitalization or Death 0.99 0.9440
AG Model with Frailty: Endpoint of HFR hospitalization 1.07 0.7584
Part 1 + Part 2 (Former Control vs. Control)
AG Model with Frailty: Endpoint of HFR hospitalization or Death 0.80 0.4512*
AG Model with Frailty: Endpoint of HFR hospitalization 0.61 0.1482*
(*P-values should be interpreted with caution because the analyses including Part 2 data were not specified before the onset of the
study and there are various sources of confounding effects which cannot be separated from the treatment effect.)
Non-Serious Adverse Device Events
There were 17 non-serious adverse device events that occurred over Part 1. There were no additional non-serious adverse device
events over Part 2 of the clinical trial. These events were rare and are well known adverse events that occur during right heart
catheterization procedures (table below).
Table 11. Non-serious Adverse Device Events Over Part 1 and Part 2
Part 1 Part 2
TREATMENT (270) CONTROL (280) ALL PATIENTS (550)
Subjects Events Subjects Events Subjects Events Subjects Events
All Patients with an Event 5 (1.9%) 6 7 (2.5%) 11 12 (2.2%) 17 0 (0%) 0
General disorders and 1 (0.4%) 1 (16.7%) 4 (1.4%) 6 (54.5%) 5 (0.9%) 7 (41.2%) 0 (0%) 0 (0%)
administration site
conditions
Catheter site bleeding 0 0 1 1 1 1 0 0
Catheter site ecchymosis 0 0 1 1 1 1 0 0
Catheter site hematoma 0 0 1 1 1 1 0 0
Chest discomfort 0 0 1 1 1 1 0 0
Chest pain 0 0 1 1 1 1 0 0
Non-cardiac chest pain 1 1 0 0 1 1 0 0
Vessel puncture site pain 0 0 1 1 1 1 0 0
Investigations 2 (0.7%) 2 (33.3%) 1 (0.4%) 1 (9.1%) 3 (0.5%) 3 (17.6%) 0 (0%) 0 (0%)
Cardiac monitoring 1 1 0 0 1 1 0 0
abnormal
Heart rate irregular 0 0 1 1 1 1 0 0
Serum creatinine increased 1 1 0 0 1 1 0 0
Respiratory, thoracic and 2 (0.7%) 2 (33.3%) 1 (0.4%) 1 (9.1%) 3 (0.5%) 3 (17.6%) 0 (0%) 0 (0%)
mediastinal disorders
49